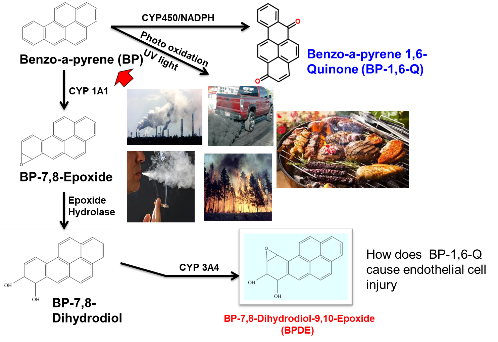
Mechanisms and cardiovascular effect of carbon nanodots
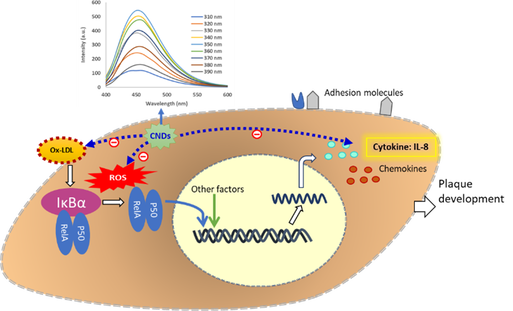
Nanoparticles, also called ultrafine particles, are small particles with a diameter between 1 and 100 nanometers (nm). Nanomaterials have received extensive attention in this century and their development and widespread applications will continue to grow in the coming years. Carbon nanodots (CNDs) are fascinating newcomers to the world of nanoparticles with sizes below 10 nm. Since their inception in 2004, carbon nanodots (CNDs) have generated much interest in the field of biomedicine, including biosensing, bioimaging and drug delivery. The photoluminescent properties of CNDs are found from an excitation wavelength of 350 nm with an emission peak at 450 nm (Belperain et al., 2021; Dunphy et al., 2021). AFM indicates that the CNDs have an average size of about 2-3 nm. Studying the biocompatibility of CNDs and their bioactivities on the cardiovascular system is crucial for improving the assessment of risk to human health by exposure to such nanoparticles. We have demonstrated that the capability of CNDs as scavengers of reactive oxygen species (ROS) including superoxide and hydroxyl radicals (Khan et al., 2021). Overproduction of ROS plays an important role in cardiovascular inflammation and its subsequent mononuclear cell adhesion to the endothelial cell (also called endothelial dysfunction), a key process in the initiation and development of atherosclerosis. We have shown that the uptake of CNDs by vascular cells is both time and concentration dependent. Our results further demonstrated that CNDs are present both intracellularly and in the mitochondria. We have examined CNDs' potential entrance into endothelial cells to understand further the action of CNDs on endothelial cells (Belperain et al., 2021). Our results showed that phenylglyoxal, a known phagocytosis inhibitor, significantly reduced the uptake of CNDs. Chlorpromazine HCL, a clathrin disruptor, also reduced CNDs uptake significantly. Our results further displayed that amlodipine, a calcium channel blocker, increased the uptake of CNDs (Belperain et al., 2021). These results suggested that the CNDs route of uptake might not only be through phagocytosis. Endothelial cells undergoing the inflammatory response produce proinflammatory cytokines and adhesion molecules. Our study has shown that CNDs can inhibit the expression of adhesion molecule and vascular inflammatory markers, including Interleukin-8 (IL-8), and interleukin 1 beta (IL-1β) (Belperain et al., 2021; Dunphy et al., 2021). Concerning the potential toxicity of CNDs, we found that C57/BL6 mice that received CNDs of 2.5 mg/Kg daily for one and two weeks did not show a significant change in body weight and serum biomarkers. No observable pathological changes were detected in various tissues. These findings provide insights on the interaction between CNDs and vascular cells involved in atherosclerosis. Our current study is focusing on evaluating toxicity and biocompatibility of CNDs both in vitro (vascular cell culture) and in in vivo (mice), investigating the effects of CNDs on inflammation-mediated atherosclerosis in vascular cells and mouse models (apolipoprotein E and low–density lipoprotein receptor knockout mice) and underlying mechanisms
Representative Publications:
- Khan, S., et al., Recent Advances in Carbon Nanodots: A Promising Nanomaterial for Biomedical Applications. Int J Mol Sci, 2021. 22(13).
- Khan, S., et al., Carbon Nanodots Inhibit Oxidized Low Density Lipoprotein-Induced Injury and Monocyte Adhesion to Endothelial Cells Through Scavenging Reactive Oxygen Species. J Biomed Nanotechnol, 2021. 17(8): p. 1654-1667.
- Dunphy, A., et al., Modulation of Macrophage Polarization by Carbon Nanodots and Elucidation of Carbon Nanodot Uptake Routes in Macrophages. Nanomaterials (Basel), 2021. 11(5).
- Belperain, S., et al., Anti-Inflammatory Effect and Cellular Uptake Mechanism of Carbon Nanodots in in Human Microvascular Endothelial Cells. Nanomaterials (Basel), 2021. 11(5).